Translate this page into:
Prevalence of malnutrition and its correlates among children coming for elective general surgical procedures in a tertiary hospital of a developing country

*Corresponding author: Dr. Olakayode O. Ogundoyin, Department of Surgery, University College Hospital, Ibadan and University College Hospital, Ibadan, Nigeria. kayogundoyin@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Adigun T, Ogundoyin OO. Prevalence of malnutrition and its correlates among children coming for elective general surgical procedures in a tertiary hospital of a developing country. Ann Med Res Pract 2020;1:1.
Abstract
Objective:
Malnutrition is a major health concern in hospitalized children. The situation in children scheduled for elective general surgery has not been clarified. The objective of the study was to assess the prevalence of malnutrition and associated factors among surgical children scheduled for elective general surgery in a typical pediatric surgical center.
Material and Methods:
This was a prospective study of 117 consecutive children scheduled for elective general surgical procedures. They had pre-operative nutritional assessment on the day of surgery using anthropometric indicators (weight for age, height for age, and body mass index for age), and malnutrition was classified based on Z score obtained using the World Health Organization Anthro and AnthroPlus Software. Operative complications and length of hospital stay were also recorded. These were analyzed using SPSS version 20. For qualitative variables, frequency and percentages were used and for quantitative variables, mean and standard deviation was used. Bivariate and multivariate logistic regression analyses were used to identify associated risk factors.
Results:
There were 85 (72.6%) boys and 32 (27.4%) girls. The mean age was 50.94 ± 43.9 with a range of 1 month–15 years. The overall prevalence of malnutrition was 46.2%. Using bivariate analysis, malnutrition was observed more in girls than in boys (P = 0.924), in children aged 1–11 months (P = 0.026), in children whose mother’s ages were 30–34 years (P = 0.045) and in children whose mothers were traders (P = 0.039). Intraoperative and post-operative complications were not associated with malnutrition (P = 0.135). The length of hospital stay was not significantly different between the malnourished and the well-nourished children. With multivariate logistic analysis, children aged 1–11 months were 4.28 times more likely to be malnourished in comparison to older children above 60 months (P = 0.034, 95% confidence interval [CI] = 1.11–16.51) and children whose mother’s ages ranged from 30 to 34 years were 3.66 times more likely to be malnourished than mothers above 40 years (P = 0.4, 95% CI = 1.01–13.19).
Conclusion:
Child’s age and mother’s age were found to be significantly associated with malnutrition. Adequate pre-operative optimization of the nutritional status of these patients is essential to good surgical outcomes.
Keywords
Anesthesia
General surgery
Malnutrition
Pediatric
Prevalence
INTRODUCTION
Malnutrition is an imbalance in nutrition, ranging from overnutrition to undernutrition.[1] It is a major public health problem in both developed and developing countries affecting all age groups but is particularly noticeable in children because they are undergoing a period of rapid growth. The cause of malnutrition in surgical patients is multifactorial; this includes deficiency in dietary intake, increased requirements related to the stage of the disease, complication of primary disease expressed in reduced absorption or excessive loss of nutrient or a combination of the above factors.[2] Patients who undergo abdominal surgery are at increased risk of malnutrition due to prolonged period of starvation[3] and those with pre-operative malnutrition are at a higher risk of developing post-operative complications, longer duration of hospital stay, and increased hospital costs.[4]
The magnitude of the problem of malnutrition in the developing countries and its association with the high rate of infant and child mortality has placed more emphasis on child and maternal nutrition.[5] The UNICEF report shows that 37% of Nigerian children below the age of 5 years are stunted, 18% are wasted, and 29% are underweight. The proportion of stunted children has declined since 2008 from 41% to 37%.[6] Malnutrition is associated with more than 50% of death (approximately 2.3 million) annually in children from developing countries.[7]
The reported prevalence of malnutrition in surgical patients is in the range of 40–67% depending on the patients’ population studied and criteria used for diagnosis.[7-9] This percentage seems to be unchanged over time and remains problematic until now. Akinbami et al.[10] reported 24.4% of underweight, 18.9% of stunting, and 13.4% of wasting in non-surgical children admitted at emergency ward in an institution in Nigeria, whereas 5 years later, Balogun and Omokhodion in another study among children with congenital heart defects in the same institution found 34.9% of wasting, 41% of stunting, and 47% underweight in children with acyanotic heart disease and in cyanotic heart disease, 29% wasting, 17.6% stunting, and 47% underweight,[11] suggesting the role of severe congenital anomalies on the nutrition and growth of these children.
Adequate information on the prevalence of malnutrition and the associated risk factors in pediatric patients undergoing elective general surgical procedures in our center is lacking. We, therefore, studied the prevalence of malnutrition and its risk factors in surgical children for elective general surgical procedures.
MATERIAL AND METHODS
This prospective, cross-sectional hospital-based study was conducted on all children planned for elective general surgical procedures in the division of pediatric surgery of our hospital from January to June 2016. Following a written and informed consent and with strict adherence to the ethical guidelines of the Declaration of Helsinki, all children under the age of 15 years scheduled for elective general surgeries were recruited into the study.
Anthropometric measurement was performed as part of pre-operative assessment of children scheduled for surgery. All children had general anesthesia with endotracheal intubation and muscle paralysis. Intraoperative monitoring included blood pressure, oxygen saturation, continuous 5 lead electrocardiogram, and temperature. At the end of the surgery, the residual neuromuscular block was reversed, and patients transferred to the recovery room. Following an Aldrete score of 8–10, patients were transferred to the ward.
A structured questionnaire was administered to obtain the socio-demographic data of the child and the caregiver. Other parameters obtained included the full blood count, intraoperative, post-operative complications, and length of hospital stay. Neonates, children scheduled for emergency surgeries, and children whose parents refused to give consent for anthropometric measurement during pre-operative assessment were excluded from the study.
The height of patients older than 2 years was measured to the nearest 1 mm using a SECA model 220 stadiometer (Medical Scales and Measuring Systems, SECA Limited, UK), whereas the body length of children aged 2 years and below was measured on a standard measuring board placed on a flat surface. Their body weight was measured to the nearest 0.1 kg using SECA 385 digital baby and infant scale (Germany) with the child barefooted but wears light cloth.
Nutritional indices which include Z-scores of weight for age, height for age, and body mass index (BMI) for age were generated from anthropometric measurement using the World Health Organization (WHO) Anthro Software Version 3.01 for patients up to 5 years of age and the WHO AnthroPlus Software Version 1.02 for patients >5 years of age. The severity of stunting, wasting, underweight, and the nutritional status was defined at Z-score ≤2 and ≤3 below the median of the reference population based on the WHO Child Growth Standards.[12]
Data collected were entered and analyzed using IBM Statistical Package for the Social Sciences Version 20 (IBM SPSS Inc., Chicago IL, USA). Quantitative variables were presented as mean and standard deviation, and qualitative variables were presented as numbers and percentages. Chi-square test was used to compare the categorical variables while bivariate and multiple logistic regressions were used to identify associated risk factors.
RESULTS
A total of 117 patients were recruited during the study period comprising 85 (72.6%) boys and 32 (27.4%) girls. The mean age was 4.25 (standard deviation [SD] ± 3.66) years with a range of 1 month–15 years. The majority (98, 83.8%) of the patients had hemoglobin concentration above 10 g/dl. The duration of hospital stays varied from 1 to 35 days with a mean of 5 days (SD ± 5.1). The majority of the caregivers were mothers of these children (115, 98%), 59 caregivers had tertiary education (50.4%), and 61 (50.4%) were traders [Table 1].
| Variables | Frequency | Percentage |
|---|---|---|
| Age (months) | ||
| 0–12 | 12 | 17.9 |
| 13–24 | 17 | 14.5 |
| 25–36 | 17 | 14.5 |
| 37–48 | 12 | 10.3 |
| ≥48 | 50 | 42.7 |
| Mother’s occupation | ||
| Trading | 61 | 52.1 |
| Teaching | 28 | 23.9 |
| Civil servant | 25 | 21.4 |
| Unemployed | 3 | 2.6 |
Overall, malnutrition was observed in 54 children (46.2%). According to weight for age, 17 (14.5%) patients were underweight only 5 (4.2%) were severely underweight with a mean Z-score of −2.91 (SD ± 1.02). According to height for age, 13 (11.1%) patients were stunted only 5 (4.3%) were severely stunted with a mean Z-score of 3.14 (SD ± 1.78), and according to the BMI for age, 37 (31.6%) children were wasted with 27 (23.1%) severely wasted with a Z-score of 11.80 ± (SD ± 49.21) [Table 2].
| Mild to moderate malnutrition (%) | Severe malnutrition (%) | Normal nutrition | Mean Z-score (SD) | ||
|---|---|---|---|---|---|
| Mean Z-score (SD) | |||||
| Weight for age (underweight) | 12 (10.3) | 5 (4.2) | 100 (85.5%) | −0.22 (0.87) | −2.91 (1.02) |
| Height for age (stunting) | 8 (6.8) | 5 (4.3) | 104 (88.9%) | 1.00 (2.54) | −3.14 (1.78) |
| Body mass index for age (thinness) | 10 (8.5) | 27 (23.1) | 80 (68.4%) | −0.08 (1.49) | −11.80 (49.21) |
Nineteen (16.2%) children were anemic; this was more in malnourished children 11 (57.9%) versus 8 (42.1 %) in well-nourished children with no significant difference (P = 0.626). Intraoperative and post-operative complications were observed in 65 (55.5%) children, some of whom had more than one complication [Figure 1], and the complications were more in malnourished children than in well-nourished children (34, 37.7% vs. 31, 52.3%; P = 0.125). Duration of a hospital stay beyond 7 days was observed in 27 (23%) children; this was more in malnourished children than in well-nourished children (14, 51.9% vs. 13, 48.1%; P = 0.498) [Table 3].
| Variable | Nourished (%) | Malnourished (%) | Total | χ2 | Df | P-value |
|---|---|---|---|---|---|---|
| Sex | ||||||
| Male | 46 (54.1) | 39(45.9) | 85 | 0.01 | 1 | 0.924 |
| Female | 17 (53.1) | 15 (46.9) | 32 | |||
| Age (months) | ||||||
| 0–12 | 6 (28.6) | 15 (71.4) | 21 | |||
| 13–24 | 10 (58.8) | 7 (41.2) | 17 | |||
| 25–36 | 7 (41.2) | 10 (58.8) | 17 | 10.37 | 5 | 0.045* |
| 37–48 | 8 (66.7) | 4 (33.3) | 12 | |||
| 49–60 | 7 (41.2) | 2 (22.2) | 9 | |||
| >60 | 25 (61.0) | 16 (39.0) | 41 | |||
| Mothers’ age (years) | ||||||
| <30 | 11 (61.1) | 7 (38.9) | 18 | |||
| 31–35 | 21 (43.8) | 27 (56.3) | 48 | 7.91 | 3 | 0.048* |
| 36–40 | 12 (46.2) | 14 (53.8) | 26 | |||
| >40 | 19 (76.0) | 6 (24.0) | 25 | |||
| Mothers’ occupation | ||||||
| Trading | 26 (42.6) | 35 (57.4) | 61 | |||
| Teaching | 16 (57.1) | 12 (42.9) | 28 | 8.35 | 3 | 0.039* |
| Civil servants | 19 (76.0) | 6 (24.0) | 25 | |||
| Unemployed | 2 (66.7) | 1 (33.3) | 3 | |||
| Hemoglobin concentration (g/dl) | ||||||
| ≤10 | 8 (42.1) | 11 (57.9) | 19 | 1.26 | 1 | 0.262 |
| >10 | 55 (56.1) | 43 (43.9) | 98 | |||
| LOS (days) | ||||||
| ≤6 | 50 (55.6) | 40 (44.4) | 90 | 0.46 | 1 | 0.498 |
| >6 | 13 (48.1) | 14 (51.9) | 27 | |||
| Complications | ||||||
| No | 32 (61.5) | 20 (38.5) | 52 | 2.229 | 1 | 0.135 |
| Yes | 31 (47.7) | 34 (52.3) | 65 |
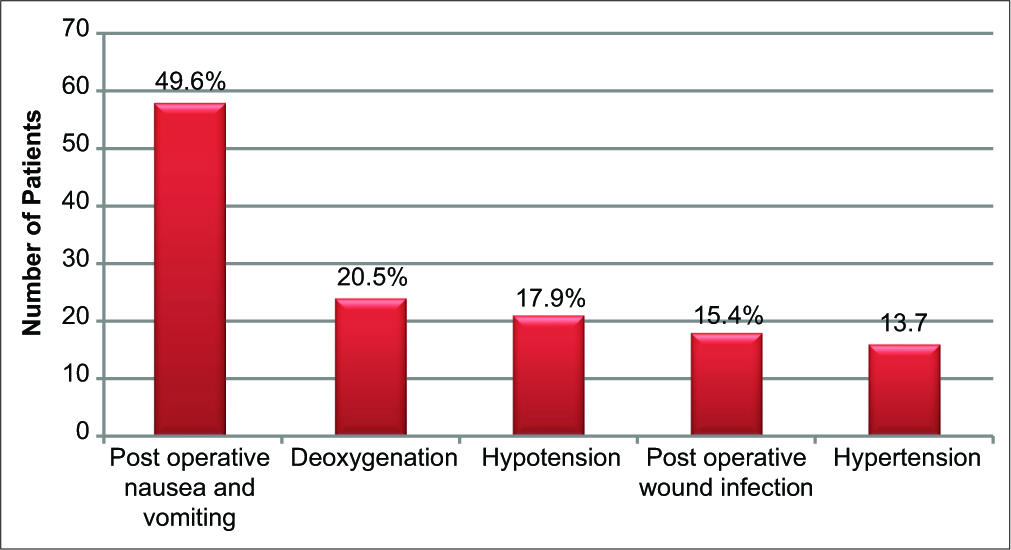
- Intraoperative and post-operative complications.
Malnutrition was most common in infants below the age of 1 year (P = 0.026), in children whose mothers’ ages ranged from 30 to 34 years (P = 0.048) and in children whose mothers were traders (P = 0.039). Children aged 1–11 months were 4.28 times more likely to be more malnourished in comparison with older children above 60 months (P = 0.034) and children whose mothers’ ages ranged from 30 to 34 years were 3.66 times more likely to be more malnourished than mothers aged 40 years and above [Table 4].
| Variable | AOR | 95% CI (AOR) | P-value | |
|---|---|---|---|---|
| Lower | Upper | |||
| Age (months) | ||||
| 0–12 | 4.28 | 1.11 | 16.51 | 0.035* |
| 13–24 | 1.70 | 0.40 | 7.20 | 0.472 |
| 25–36 | 1.42 | 0.36 | 5.60 | 0.615 |
| 37–48 | 0.78 | 0.17 | 3.58 | 0.750 |
| 49–60 | 0.30 | 0.05 | 1.83 | 0.191 |
| >60 | 1 | |||
| Mothers’ age (years) | ||||
| <30 | 1.59 | 0.31 | 8.03 | 0.576 |
| 31–35 | 3.66 | 1.01 | 13.19 | 0.047* |
| 36–40 | 3.58 | 0.96 | 13.33 | 0.057 |
| >40 | 1 | |||
| Mothers’ occupation | ||||
| Trading | 3.56 | 0.20 | 62.67 | 0.385 |
| Teaching | 0.99 | 0.05 | 19.05 | 0.992 |
| Civil servants | 0.83 | 0.04 | 16.25 | 0.901 |
| Unemployed | 1 | |||
| Complications | ||||
| No | 1.47 | 0.61 | 3.52 | 0.393 |
| Yes | 1 | |||
AOR: Adjusted odds ratio, CI: Confidence interval
DISCUSSION
Nutritional status is one of the indices of assessment of the general health status of children. Although observed the prevalence of malnutrition was lower in comparison to the result of the Nigeria demographic and health survey,[8] it is higher than reported prevalence from Germany (6.1%), France (11%), and Brazil (6.9%).[12-14] This lower prevalence rate may be related to the difference in geographical location, and population studied as these countries are high-income countries with better health-care delivery systems and per capita income.
Documented prevalence rates among different specialties of surgeries revealed higher prevalence rates; Rojratsirikul et al. reported 46% of malnutrition among pediatric surgical patients in 78 children aged 2 months–16 years,[15] Secker and Jeejeebhoy reported 51% prevalence of malnutrition in 175 children with major thoracic or abdominal surgery[16] and Okoromah et al. reported 90.4% of malnutrition among pediatric patients with congenital heart disease scheduled for corrective heart surgery,[17] whereas a study conducted on adult general surgical patients identified a lower prevalence of 6.1% prevalence.[18] These relatively high prevalence rates suggest the role of associated severe congenital malformations and malnutrition as causes of failure to thrive. Different prevalence rates of 19% and 24.5% were also documented in non-surgical diseases by Joosten et al.[19] and Hendricks et al.,[20] respectively.
It was observed that there was a significant association between the ages of the children and nutritional status. Children aged 1 year and below were 4.28 times more likely to develop malnutrition than older age groups. Similarly, a hospital-based study in Brazil revealed that children aged 1 year or less were 2.31 times more likely to suffer malnutrition than children above 1 year of age.[21] In contrast, Rocha et al. reported a higher level of malnutrition among the older age group (23–59 months) than the younger age group (2–11 months).[22] The higher prevalence rate of malnutrition among younger pediatric surgical patients may be due to congenital anomalies (gastrointestinal) that usually cause failure to thrive as they require more energy intake for growth and development. Other possible causes of failure to thrive in patients with associated congenital abnormalities as suggested by the previous growth and nutritional study[23] include inadequate caloric intake, intestinal malabsorption, increased energy expenditure, and decreased splanchnic blood flow.
There was no significant association between the sex of the children and malnutrition; nevertheless, it was observed that girls suffered more malnutrition than boys. Perhaps a larger sample size with more girls recruited for the study may be more appropriate to make a concrete conclusion.
There was a significant association between maternal age and the nutritional status of the children; this is in contrast to an earlier study that showed an association between child stunting at 2 years of age and young maternal age.[24] They attributed the finding to low socio-economic factors in this group of mothers. Although the mothers of the patients studied were older, their low socio-economic status may be a factor.
Poor nutrition in hospitalized patients undergoing elective surgery in an adult is associated with increased post- operative infection, which may lead to longer hospital stay,[25] an increase in the incidence of post-operative infection was however not observed in this series.
CONCLUSION
The prevalence of malnutrition in pediatric surgical patients scheduled for elective general surgery is high in our hospital. This has impacted greatly on the outcome of surgery, as the attendant high morbidity would increase the duration of hospital stay after surgical management. Adequate optimization of the nutritional status of these patients is essential to good outcomes. Therefore, we suggest that surgeons, the government, non-government organizations, and other relevant stakeholders should work together to make health-care services affordable and accessible to enhance early diagnosis, adequate preoperative optimization of the nutritional status, prompt treatment, and proper rehabilitation of the children especially the ones with associated congenital malformations.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Prognostic impact of disease-related malnutrition. Clin Nutr. 2008;27:5-15.
- [CrossRef] [PubMed] [Google Scholar]
- A rational approach to nutritional assessment. Clin Nutr. 2008;27:706-16.
- [CrossRef] [PubMed] [Google Scholar]
- Nutritional screening tools and prediction of malnutrition, incidence in major abdominal surgery patients at a tertiary hospital in Bangkok, Thailand. Mahdol Univ J Pharm Sci. 2013;40:26-34.
- [Google Scholar]
- Economic impact of malnutrition: A model system for hospitalized patients. JPEN J Parenter Enteral Nutr. 1988;12:371-6.
- [CrossRef] [PubMed] [Google Scholar]
- The State of the World's Children. Vol 2005. New York: The United Nations Children's Fund;
- [Google Scholar]
- Nigeria Demographic and Health Survey 1999. In: The Commission. Abuja, Nigeria: National Population Commission; 2000.
- [Google Scholar]
- Prevalence of hospital malnutrition in Latin America. The multicentre ELAN study. Nutrition. 2003;19:823-5.
- [CrossRef] [Google Scholar]
- Prevalence of malnutrition in surgical patients: Evaluation of nutritional support and documentation. Clin Nutr. 1999;18:141-7.
- [CrossRef] [Google Scholar]
- Malnutrition in surgical patients. An unrecognised problem. Lancet. 1977;1:689-92.
- [CrossRef] [Google Scholar]
- Body mass composition: A predictor of admission outcomes among hospitalized Nigerian under 5 children. Asia Pac J Clin Nutr. 2010;19:295-300.
- [Google Scholar]
- Nutritional profiles and selected parental factors among children with congenital heart diseases in Ibadan, Nigeria. Nig J Cardiol. 2015;12:89.
- [CrossRef] [Google Scholar]
- WHO child growth standards based on length/height, weight and age. Acta Paediatr Suppl. 2006;450:76-85.
- [CrossRef] [Google Scholar]
- Prevalence of malnutrition in paediatric hospital patients. Clin Nutr. 2008;27:72-6.
- [CrossRef] [PubMed] [Google Scholar]
- Malnutrition screening in hospitalized children: Influence of the hospital unit on its management. Arch Pediatr. 2005;12:1226-31.
- [CrossRef] [PubMed] [Google Scholar]
- Application of subjective global assessment as a screening tool for malnutrition in pediatric surgical patients. J Med Assoc Thai. 2004;87:939-46.
- [Google Scholar]
- Subjective global assessment for children. Am J Clin Nutr. 2007;85:1083-9.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence, profile and predictors of malnutrition in children with congenital heart defects: A case-control observational study. Arch Dis Child. 2011;96:354-60.
- [CrossRef] [PubMed] [Google Scholar]
- The efficacy of nutrition risk screening-2002 (NRS-2002) to decide on the nutritional support in general surgery patients. Bratisl Lek Listy. 2009;110:290-2.
- [Google Scholar]
- Prevalence of malnutrition in pediatric hospital patients. Curr Opin Pediatr. 2008;20:590-6.
- [CrossRef] [PubMed] [Google Scholar]
- Malnutrition in hospitalized pediatric patients. Current prevalence. Arch Pediatr Adolesc Med. 1995;149:1118-22.
- [CrossRef] [PubMed] [Google Scholar]
- Anthropometric evaluation, risk factors for malnutrition, and nutritional therapy for children in teaching hospitals in Brazil. J Pediatr (Rio J). 2009;85:223-8.
- [CrossRef] [Google Scholar]
- The effects of hospitalization on the nutritional status of children. J Pediatr (Rio J). 2006;82:70-4.
- [CrossRef] [PubMed] [Google Scholar]
- The influence of nutritional status on complications after major intraabdominal surgery. J Am Coll Nutr. 2004;23:227-32.
- [CrossRef] [PubMed] [Google Scholar]
- Association maternal age at childbirth and child and adult outcomes in the offering; a prospective study in five low income and middle income countries (COHORT collaboration) Lancet Glob Health. 2015;3:e366-77.
- [CrossRef] [Google Scholar]
- Is malnutrition still a risk factor of postoperative complications in gastric cancer surgery? Clin Nutr. 2008;27:398-407.
- [CrossRef] [PubMed] [Google Scholar]






